Mass periodic table definition chemistry2/16/2024  
Meet the Elements by They Might be Giants.The Elements by Tom Lehrer with animation by Mike Stanfill.Since they were working with actual samples of elements such as copper, mercury, sulfur, etc., they called it the periodic table of the elements. The people who developed the periodic table could have called it the Periodic Table of the Atoms but they did not have a firm understanding of atoms at that time. This piece of pure carbon is a sample of the element carbon. For instance, a piece of pure carbon is made up of only carbon atoms. 
An element is a substance made up of all the same type of atom. Explain to students that an atom is the smallest particle or “building block” of a substance. Note : It is often confusing for students to see the terms “atom” and “element” used interchangeably as if they are the same thing. The 100 or so atoms of the periodic table, in different combinations, make up millions of different substances. It’s kind of like the alphabet in which only 26 letters, in different combinations, make up many thousands of words. The periodic table shows all the atoms that everything in the known universe is made from. 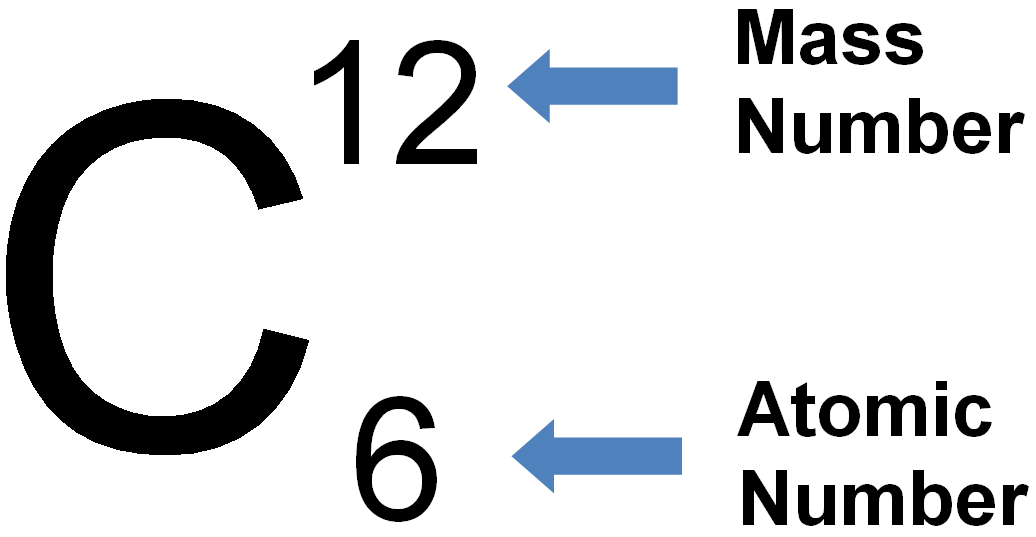
Explain that each box contains information about a different atom. Tell students that this is the periodic table. Lessons 2 and 3 both use the 20 sheets of atom description cards – one sheet for each element. They will also be able to explain the meaning of atomic number and atomic mass. 
Students will identify different atoms by the number of protons in the nucleus and realize that the number of electrons equals the number of protons in a neutral atom. Students will then watch several videos of some interesting chemical reactions involving some of these elements. They will try to correctly match cards with information about an element to each of the first 20 elements. Students will focus on the first 20 elements. They will be introduced to the basic information given for the elements in most periodic tables: the name, symbol, atomic number, and atomic mass for each element. Students will begin to look closely at the periodic table. The atoms in the periodic table are arranged to show characteristics and relationships between atoms and groups of atoms.The atomic mass of an element is the average mass of the different isotopes of the element.Atoms of the same element with different numbers of neutrons are called “isotopes” of that element.Different atoms of the same element can have a different number of neutrons.The number of electrons surrounding the nucleus of an atom is equal to the number of protons in its nucleus.The atomic number of an atom is equal to the number of protons in its nucleus.An element is a substance made up of only one type of atom.The periodic table is a chart containing information about the atoms that make up all matter.To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. This is repeated until there is a term for each isotope. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average. = 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
